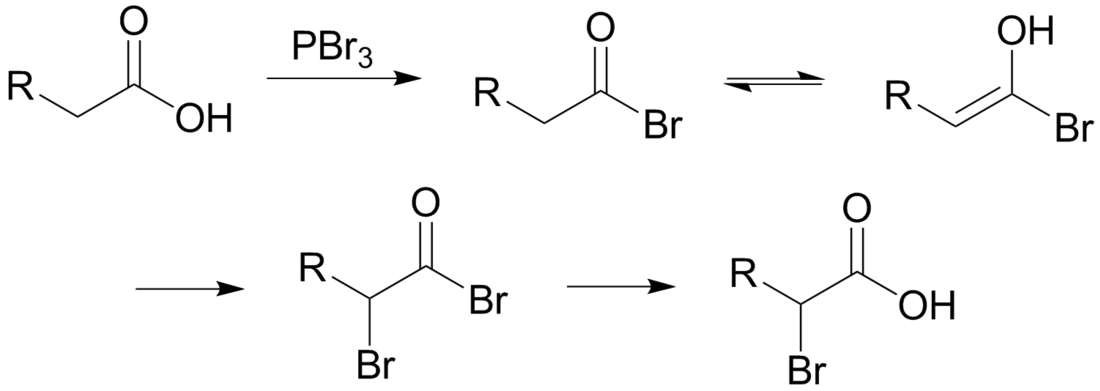
The Hell–Volhard–Zelinsky halogenation reaction is a chemical transformation that transforms an alkyl carboxylic acid to the α-bromo derivative. It is a specialized and rare kind of halogenation.[1][2]
| Hell–Volhard–Zelinsky halogenation | |
|---|---|
| Named after | Carl Magnus von Hell Jacob Volhard Nikolay Zelinsky |
| Reaction type | Substitution reaction |
| Identifiers | |
| Organic Chemistry Portal | hell-volhard-zelinsky-reaction |
Examples
An example of the Hell–Volhard–Zelinsky reaction can be seen in the preparation of alanine from propionic acid. In the first step, a combination of bromine and phosphorus tribromide (catalyst) is used in the Hell–Volhard–Zelinsky reaction to prepare 2-bromopropionic acid,[3] which in the second step is converted to a racemic mixture of the amino acid product by ammonolysis.[4][5]
Mechanism
The reaction is initiated by addition of a catalytic amount of PBr3, after which one molar equivalent of Br2 is added. PBr3 converts the carboxylic OH to the acyl bromide. The acyl bromide tautomerizes to an enol, which reacts with the Br2 to brominate at the α position.In neutral to slightly acidic aqueous solution, hydrolysis of the α-bromo acyl bromide occurs spontaneously, yielding the α-bromo carboxylic acid. If an aqueous solution is desirable, a full molar equivalent of PBr3 must be used as the catalytic chain is disrupted.
If little nucleophilic solvent is present, reaction of the α-bromo acyl bromide with the carboxylic acid yields the α-bromo carboxylic acid and regenerates the acyl bromide intermediate. In practice a molar equivalent of PBr3 is often used anyway to overcome the slow reaction kinetics.
The mechanism for the exchange between an alkanoyl bromide and a carboxylic acid is below. The α-bromoalkanoyl bromide has a strongly electrophilic carbonyl carbon because of the electron-withdrawing effects of the two bromides.
By quenching the reaction with an alcohol, instead of water, the α-bromo ester can be obtained.
History
The reaction is named after the German chemists Carl Magnus von Hell (1849–1926) and Jacob Volhard (1834–1910) and the Russian chemist Nikolay Zelinsky (1861–1953).[6][7][8]
See also
References
Wikiwand in your browser!
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.