Preprint
Article
Effects of Sand Burial Depth on Bivalve Species Predation by the Invasive Blue Crab, Callinectes sapidus
Altmetrics
Downloads
104
Views
47
Comments
0
A peer-reviewed article of this preprint also exists.
This version is not peer-reviewed
Submitted:
02 May 2024
Posted:
03 May 2024
You are already at the latest version
Alerts
Abstract
In the Ebro Delta, the abundance of burrowing bivalves has dramatically decreased, with the blue crab, Callinectes sapidus, being blamed by shellfish collectors. Trends from 2010 evidence a decrease in the capture of clams (Ruditapes spp.) before 2016 (start of blue crab fisheries), although further decline of both clam and cockle (Cerastoderma glaucum) occurred in 2018. In contrast, captures of razor clam (Ensis siliqua) have increased by 3.6-fold since 2016. Predation risk for these taxa with contrasting burrowing capacities (1.7 ± 0.3cm, 0.4 ± 0.2cm, and 26.3 ± 0.1cm, respectively), was assessed using predation preference (N= 5 tanks; 5 individuals of each species) and no-choice experiments (N= 5 tanks; 15 individuals) in the absence and in the presence of sand. Results showed that in the absence of sand, the razor clam, was fully preyed in 24h, clams in 96h, and cockles reached 60% after 144h. Conversely, when sand was present, only 4% of razor clams were predated, while clams and cockles reached 60-100% in 120-144h. No-choice results featured similar patterns depending on substrate availability. Overall, clams and cockles appear to be greatly vulnerable to blue crab predation, whereas the razor clam may escape thanks to its deeper burrowing capacity.
Keywords:
Subject: Biology and Life Sciences - Aquatic Science
1. Introduction
The blue Atlantic crab, Callinectes sapidus Rathburn, 1896, is a marine decapod native to the western Atlantic region, from Maine to the Río de la Plata [1]. The species features euryhaline and eurythermal capabilities that allow it to colonize both coastal and freshwater habitats, coupled with high fecundity and aggressive behavior [2,3]. Since its accidental introduction to the Eastern Mediterranean Sea in 1948 blue crab abundances have been progressively increased [4] impacting natural ecosystems and local fisheries [3,5,6].
Although the blue crab is regarded as a generalist omnivorous consumer feeding on a variety of food resources depending on availability and size [7,8], bivalves appear to be a favorite prey item for juveniles and adult individuals [9,10], thus becoming one of the most vulnerable taxa to predation. Previous experimental research with non-burying bivalves has reported important predation effects associated to factors such as prey density, attachment strength, shell size and hardness, and as well as to distinctive nutritional features [11,12,13,14] that point to a large variability in predation vulnerability across species. A particular decisive aspect is the arrival to a critical upper threshold size – from >40 mm in the hard clam, Mercenaria mercenaria [11] to 80-90 mm for the ribbed mussel, Geukensia demissa [13] – at which not even large crabs can consume the prey, but such size might not be attained [14]. However, for burying bivalves, the protection of substrate, rather than prey size, might reduce predation rates, depending on the relative burying capacities of both predator and prey and the sensory abilities to detect buried preys at various densities [15,16]. Blue crabs are tactile feeders, capable of extracting bivalves up to a maximum burial depth of ca. 10–15 cm of substrate [17]. In contrast, burying ranges of bivalves might widely differ across species, from sub-superficial layers in Donax spp., Ruditapes spp. or Cerastoderma spp. [18] to over 1 m in razor clams [19], which could provide enhanced chances to escape from predation to species with greater digging capacities.
In the last two decades the blue crab has experienced an expansion of towards the Western Mediterranean basin [5]. Specifically, in the Ebro Delta, the species was first reported in 2012 from the Tancada Lagoon [20], and since then, it has become increasingly abundant [21,22]. Alongside, the remaining local populations of Cerastoderma spp. and Ruditapes spp. in the Alfacs and Fangar Bays have virtually disappeared over the last years. Also, the hatchery production of the Japanese clam Ruditapes philippinarum in the Fangar Bay has closed because of high predation losses (Vongole 2000 S.L., pers. communication from the manager, P. López to I. Gairin), and presently, the only bivalve species that seems to be captured in large abundance in shallow invaded blue crab habitats is the razor clam, Ensis siliqua, according to data provided by the Catalan Research Institute for the Governance of the Sea (ICATMAR).
In this context, the objective of the present work is two-fold. First, we conducted several manipulative experiments (multiple choice and no choice) aimed to assess preferential predation rates across the three taxa commonly sharing habitat with the blue crab, and with higher commercial interest (Cerastoderma spp., Ruditapes spp., and E. siliqua). Second, we aimed to evaluate capture data from 2010 to 2023 from the different local fisheries across the Ebro Delta in order to assess possible patterns in bivalve trends associated to the increasing captures of blue crab, as proxies of local abundances.
2. Materials and Methods
2.1. Collection of Predators and Prey Items
Blue crab individuals were bought alive from the fishermen’s association of Riumar located in the town of Deltebre, close to the mouth of the Ebro River. Only males were used throughout the experiments in order to avoid sex-related differences in claw morphology leading to possible variability in predation efficiency [12]. Individuals with a medium size (150–250 g WW) were selected as the most representative fishery size and replaced after each experimental trial. Crabs were transported to IRTA facilities 24 h before experiments, in order to allow for acclimation of individuals and ensure non-feeding conditions during that period.
Bivalve species from local catches in the Ebro Delta were bought alive at the Riumar fishermen’s association (E. siliqua, Ruditapes spp., and C. glaucum). Individuals (including shells) were selected with comparable weights (Ruditapes spp.: 14.7 ± 0.3 g WW; C. glaucum: 14.5 ± 0.3 g WW; and E. siliqua: 14.3 ± 0.2 g WW) in order to avoid possible size effects in predation preferences. Individuals of each species were also brought to our facilities 24 h before each experiment, in order to allow for natural burying depths in sand substrate trials (see below). Additional individuals of each species (N= 5) were also used to assess burying variability across species using a 2 L glass graduated measuring cylinder.
2.2. Predation Experiments
Multiple choice experiments were first conducted with the three species of bivalves in order to assess predation preferences in the presence and in the absence of sand substrate. For each trial, N= 5 plastic tanks of 120 L, 50 cm diameter, and 100 cm height were used to host one blue crab and N= 5 individuals of each species (i.e., a total of 15 bivalves per tank). In no choice experiments, N= 5 tanks hosting one blue crab individual were used for each bivalve species (N= 15 per tank) also in the presence and in the absence of sand. In all experiments, daily predation rates were visually monitored for empty shells or possible fragments, until at least two of the three species were fully consumed or destroyed (usually 144 h).
Individuals of Ruditapes spp. and C. glaucum were usually opened by exerting pressure with the claw until both valves were slightly ajar and then prying until the abductor muscle was torn (Figure 1a,b). In contrast, predation of E. siliqua was facilitated by a very thin shell, which leaded to easy fragmentation and rapid death of individuals during manipulation with possible presence of unconsumed flesh remains (see Figure 1c).
2.3. Fisheries Trends
Fisheries landings communicated daily from fish markets to the Catalonian government were facilitated by ICATMAR. We selected data from 2010 to 2023 in order to assess patterns before and after the beginning of blue crab fisheries in August 2016, after the species started to become abundant and fishing permission was granted by not including C. sapidus in the Spanish invasive species list [21]. Selected bivalve species included those used in the predation experiments (Ruditapes spp., C. glaucum and E. siliqua) and the wedge clam (Donax spp.) as a negative control, since it mostly occurs in exposed open sea areas where the blue crab is not abundant [23]. Other locally present commercial species of Venus clams (Venus casina, V. verrucosa, and Chamelea gallina), were pooled together to assess general effects on the remaining bivalve community.
Fisheries data were expressed as annual captures in kg and not in CPUEs, because of inadequate registration of effort for bivalves collected with manual methods (fictional groupings of shellfish collectors instead of individuals) by fishermen’s guilds before 2019 (personal communication from ICATMAR to P. Prado). Yet, captures from 2010 to 2023 for each taxa were still significantly associated to available reports of effort (0.76≤ R2≤ 0.96 for bivalves and R2= 0.98 for blue crab) were considered as an adequate proxy of field abundances.
2.4. Data Analysis
Experimental data on predation rates in number of predated individuals were transformed to percent cumulative predation for data analyses purposes. Results were analyzed with two way repeated-measures analysis of variance (RM-ANOVA) using a generalized linear model followed by Tukey post hoc testing to determine significant groupings. The validity of the F-statistic used in the RM-ANOVA was examined by performing Mauchly’s test of sphericity. Since sphericity could not be assumed, the less conservative Huynh-Feldt criteria was applied. For all tests, a p-value of <0.05 was considered statistically significant.
The possible association between blue crab captures and those of the different bivalve taxa for the 2016-2023 period (N= 8) was investigated with regression analyses.
3. Results
3.1. Multiple Choice Experiments
RM-ANOVA showed significant time effects, with increasing cumulative prey predation along the experiment (144 h> 120 h> 96 h> 72 h= 48 h> 24 h> 0 h) (Table 1a; Figure 2a,b). However, predation rates over time were uneven across species, with Ruditapes spp. being preyed at a quicker pace than C. glaucum both in the presence and the absence of sand, whereas predation on E. siliqua was the fastest in the absence of substrate and the slowest in the presence of sand (Table 1a; Figure 2a,b).
There were also significant effects of substrate, species and their interactions. The presence of sand substrate significantly decreased the overall predation compared to bare substrate (68 ± 12.2% vs. 86.7 ± 6.7%, respectively). For species, predation rates were significantly higher in Ruditapes spp. (100% in all cases), followed by C. glaucum (80 ± 9.4%) and E. siliqua (52 ± 16.1%). The presence of sand substrate coupled with a large burial depth capacity (mean of 26.3 ± 4.1 cm) was a key factor for the survival E. siliqua (4 ± 4% vs. 100% predation, respectively in the presence and in the absence of sand) (Figure 2a,b). In contrast, Ruditapes spp. and C. glaucum featuring a very shallow burial (1.7 ± 0.3 and 0.4 ± 0.2 cm, respectively) showed, either the same or even more predation in the presence of sand.
3.2. No Choice Experiments
RM-ANOVA also showed increasing cumulative predation over the experimental period (144 h= 120 h= 96 h= 72 h≥ 48 h> 24 h> 0h), with significant interaction effects with substrate and species (Table 1b; Figure 2c,d). Compared to multiple choice results, when substrate was present, blue crabs predated individuals of C. glaucum at a faster rate than those of Ruditapes spp., whereas a similar lower predation pattern was observed for E. siliqua. In contrast, in the absence of substrate and other bivalves, all prey species were preyed at a similar high rate at all experimental times (Figure 2c,d).
The presence of sand also resulted in decreased predation compared to bare substrate (72.4 ± 10.6% vs. 100% predation). Besides, among species there was significantly higher predation of C. glaucum and Ruditapes spp. (100% in both of them) than E. siliqua (58.7 ± 14.2%). As in multiple choice results, the presence of sand was a central factor for the survival of E. siliqua (17.3 ± 7.5% vs. 100% predation), but showed no effects for the other taxa (Figure 2c,d).
3.3. Fisheries Trends
The capture of cockles has dramatically decreased from ca. 12,000-14,000 kg in 2010-2011 to only 1755 kg in 2023. Besides, an abrupt decline with no further recover was observed in 2018, shortly after an increase of blue crab captures (Figure 3), resulting in a significant association between both taxa (R2= 0.664, df= 7, F= 11.86, p= 0.013). For clams, 2010 abundances were already low (3537,2 kg) and have been jaggedly declining until reaching only 59,3 kg in 2023 but no significant association with blue crab captures was observed due to strong interannual variability (R2= 0.02, df= 7, F= 0.538, p= 0.490). In contrast, the abundance of razor clam has unevenly increased from 31,879 kg in 2010 to up to 110,488 kg in 2023, particularly after 2019 (Figure 3), but no significant association was either observed (R2= 0.305, df= 7, F= 4.084, p= 0.089).
The wedge clam displayed an extremely jagged pattern of captures over the years previously and after the arrival of blue crab, featuring similar values in 2010 (50,594 kg) and 2023 (47,230 kg) (R2= 0.283, df= 7, F= 3.543, p= 0.096). For the pooled Venus clam taxa, there was a major peak in 2013-2014 with values increasing from ca. 3,500-7,000 kg to up to 41,779 kg and then decreasing again to similar values in 2015 (Figure3). Later, in 2022-2023, captures increase again to the 10,000-20,000 kg range (R2= 0.135, df= 7, F= 0.860, p= 0.389).
4. Discussion
4.1. Patterns of Experimental Predation
Our results are in line with the findings of other studies reporting declining crab predation with increasing burying depth for other bivalve species such as Paphies ventricosa [15] and Macoma balthica [24] buried at different depths, and further demonstrate that it can act as a refuge from predation. However, we provide additional evidence that burial depth might be responsible for distinctive predation rates across coexisting bivalve species and contribute to shaping abundance patterns in invaded Mediterranean ecosystems.
We show that the razor clam, E. siliqua, featuring a burrowing depth range between 17.5 to 40 cm, is not efficiently extracted by blue crab, with predation rates reaching maximum values of only 20-33% across the experiments. This burial range is lower than that described in the natural media (up to 1 m; [19]) but still exceeds the critical 10–15 cm depth indicated for blue crab by Seitz et al. [17], suggesting that E. siliqua can still be exposed to certain predation, which might be related to a less evasive burrowing behavior following disturbance compared to other species in the genus [25]. In contrasts, in the absence of substrate, predation on razor clam reached similar or higher rates than the other species, possibly because of the effect of enhanced shell fragility on prey vulnerability [14]. Besides, in the absence of substrate, predation rates in multiple-choice experiments were also much higher on razor clam, pointing to a preferential attack, possibly because of manipulative easiness [14,26], since prey availability was kept alike in all experiments (ca. 76.4 ind. · m−2). Other factors such as enhanced palatability or profitability alone do not seem to be a clear predictor of blue crab preference (see also Ebersole and Kennedy [27]) for razor clam, since in the absence of substrate, the species was quickly attacked but largely unconsumed (Figure 1), while this pattern was not observed for the other two bivalves.
Clams and cockles both featured similarly low burrowing capacities (<2 cm) lying within the crab excavating ability [17], and the presence of substrate did not result in enhanced protection from predation. However, predation rates during multiple-choice experiments were consistently higher and occurred at a faster pace in clams than in cockles (up to 100% vs. 60% in the absence of substrate), suggesting additional preferential predation. Microhardness (fracture toughness of the material) tests conducted on the shells of several Mediterranean species evidenced ca. 26% higher values on Cerastoderma than in Ruditapes [28], which could account for observed differences in predation. Alternatively, cockles might also feature significantly lower protein and lipid contents than coexisting clams [29], and could be less preferred for nutritional reasons. Only when no-choice was available, predation on C. glaucum was significantly increased (up to a 40%) and reached similar values to Ruditapes spp.
4.2. Bivalves’ Fisheries Patterns
Experimental patterns of blue crab predation, were in accordance with trends in capture fisheries obtained from ICATMAR. The extraction of razor-clam (mostly E. siliqua, and to a lesser extent Solen marginatus) has been outstandingly increased by 3.6-fold since the first captures of blue crab in 2016 [21,22] as a result of an increasing fishing effort (R2= 0.554), which might partly obscure predation effects. In contrast, the extraction of Cerastoderma spp. from natural banks has decreased from over 12,000 kg to less than 2,000 kg since the arrival of blue crab, coupled with decreasing capture effort during this period. Besides, local populations in the Ebro have been undergoing repeated infections of the parasite Marteilia cochillia [30] with an unknown evolution forecasting due to a lack of monitoring. Also, there is an important small-scale fishery using mechanized dredges to extract clams along the Catalan coast [31], which has been shown a significant negative effect on the subsequent settlement of the species [32]. For clams (Ruditapes spp.), a large decrease in captures was observed in 2018 (by ca. 12-fold of the average of the previous 7 years), only two years after increasing abundances made of blue crab a commercial fishing target [21] and captures have reached minimums of only 2.3 and 59.3 kg in 2022-2023. In this case, predation appears to have played a major role that forced the closure of the last remaining cultivation company (Vongole 2000 S.L., pers. communication from the manager, P. López to I. Gairin). However, the production of Ruditapes spp. in shallow areas of the Alfacs and Fangar Bays reached values of over 250 tons in the late 90s [33] and crop values from 2010 to 2015 were also considerably higher (an average of ca. 1,500 kg). Since over 75% of the Ebro Delta surface is devoted to rice cultivation, bays have been greatly exposed to agricultural pollution and feature anoxic sediments (P. Prado, personal observ.), that impact commercially exploited shallow natural banks and also affecting suspended cultures of mussels and oysters [34]. The extraction of other species of Venus clams, was dominated by the thick-ridged venus (V. casina; over 90% of total after 2015), and to a lesser extent by the striped venus clam (C. gallina) and the warty venus (V. verrucosa), that occur in open waters of the Ebro Delta [31,35]. In particular, the thick-ridged venus, is found at depths over 120 m [36] that the blue crab does not reach, and seems to have become an increasing alternative resource compared to other heavily exploited Venus clams such as the striped venus [31]. In shallow exposed open sea areas that are neither frequented by the blue crab the abundance of wedge clam (Donax spp.) is subjected to large interannual variability, possibly due to the effect of fishing pressure and/ or natural density dependent processes affecting spawning and recruitment [23,31]. In fact, recent research in open waters of the Ebro Delta, has found that bivalves are a minor component of the blue crab diet, which appears to be feeding at much higher trophic levels [37].
5. Conclusions
The evidence presented herein shows that the burrowing capacity inherent to each bivalve species, is a major factor determining predation rates of blue crab; species inhabiting shallow sands (<2 cm) such as Ruditapes spp. and Cerastocerma spp. being ca. 6 to 25 times more vulnerable than razor clam capable of accessing to depth sands (>25 cm). Capture data from local fisheries rates as a proxy of field abundance of the different bivalve species appear to be coherent with experimental top-down patterns, razor clam being the only taxa that is still being captured in large quantities. However, the undergoing of other factors such as fishing pressure, pollution, or diseases [30,31,33] might have also greatly contributed to the collapse of cockle and clam captures in the Ebro Delta. Overall, urgent management measures are needed to recover the missing prosperity and diversity of bivalve resources in the region during the 90s and 00s [33].
Author Contributions
Conceptualization, P.P., and S.F.; Data curation, P.P; Formal analysis, P.P., and S.F.; Investigation, P.P.; Methodology, P.P., and I.G..; Study design, P.P., and S.F.; Data analysis, P.P.; Resources, P.P., and S.F.; Supervision, P.P.; Visualization, P.P., I.G., and S.F.; Writing—original draft, P.P.; Writing—review and editing, S.F., and I.G. All authors have read and agreed to the published version of the manuscript.
Funding
Predation experiments were supported by the Spanish Government (Ministry of Science and Technology) under the ECESIS project (PID2020-118476RR-C21) to P. Prado and S. Falco.
Informed Consent Statement
Not applicable.
Data Availability Statement
We encourage all authors of articles published in MDPI journals to share their research data. In this section, please provide details regarding where data supporting reported results can be found, including links to publicly archived datasets analyzed or generated during the study. Where no new data were created, or where data is unavailable due to privacy or ethical restrictions, a statement is still required. Suggested Data Availability Statements are available in section “MDPI Research Data Policies” at https://www.mdpi.com/ethics.
Acknowledgments
P. Prado was contracted under the INIA-CCAA research program for postdoctoral incorporation from the Spanish National Institute for Agricultural and Food Research and Technology (INIA). We thank C. Alcaraz for advise with the experimental design of the predation experiments. We are very grateful to the ICATMAR institution for providing the fisheries data on blue crab and bivalve species used in this study.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Hill, J.; Fowler, D.L.; Avyle, M.V. Species profiles: Life histories and environmental requirements of coastal fishes and invertebrates (Mid-Atlantic) – Blue crab. U.S. Fish and Widlife Service Biological Report. 1989, 82, 18 pp. [Google Scholar]
- Hines, A.H. Ecology of juvenile and adult blue crabs: summary of discussion of research themes and directions. Bull. Mar. Sci. 2003, 72, 423–433. [Google Scholar]
- Mancinelli, G.; Carrozzo, L.; Marini, G.; Costantini, M.L.; Rossi, L.; Pinna, M. Occurrence of the Atlantic blue crab Callinectes sapidus (Decapoda, Brachyura, Portunidae) in two Mediterranean coastal habitats: temporary visitor or permanent resident? Estuar. Coast. Shelf Sci. 2013, 135, 46–56. [Google Scholar] [CrossRef]
- Nehring, S. Invasion history and success of the American blue crab Callinectes sapidus in European and adjacent waters. In Galil, B.S., Clark, P.F., Carlton, J.T. (Eds.), In the wrong place–alien marine crustaceans: distribution, biology and impacts; Invading Nature-Springer Series 6: Berlin, Germany, 2011; pp. 607–624. [Google Scholar]
- Mancinelli, G.; Chainho, P.; Cilenti, L.; Falco, S.; Kapiris, K.; Katselis, G.; Ribeiro, F. The Atlantic blue crab Callinectes sapidus in southern European coastal waters: distribution, impact and prospective invasion management strategies. Mar. Poll. Bull. 2017, 119, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Clavero, M.; Franch, N.; Bernardo-Madrid, R.; López, V.; Abelló, P.; Queral, J. M.; Mancinelli, G. Severe, rapid and widespread impacts of an Atlantic blue crab invasion. Mar. Poll. Bull. 2022, 176, 113479. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.M.; Weissburg, M.J. Habitat complexity and predator size mediate interactions between intraguild blue crab predators and mud crab prey in oyster reefs. Mar. Ecol. Progr. Ser. 2013, 488, 209–219. [Google Scholar] [CrossRef]
- Prado, P.; Ibáñez, C.; Chen, L.; Caiola, N. Feeding habits and short-term mobility patterns of blue crab, Callinectes sapidus, across invaded habitats of the Ebro Delta subjected to contrasting salinity. Estuar. Coast. 2022, 45, 839–855. [Google Scholar] [CrossRef]
- Miller, R.E.; Sulkin, S.D.; Lippson, R.L. Composition and seasonal abundance of the blue crab, Callinectes sapidus Rathbun, in the Chesapeake and Delaware Canal and adjacent waters. Chesapeake Sci. 1975, 16, 27–31. [Google Scholar] [CrossRef]
- Laughlin, R.A. Feeding habits of the blue crab, Callinectes sapidus Rathbun, in the Apalachicola estuary, Florida. Bull. Mar. Sci. 1982, 32, 807–822. [Google Scholar]
- Arnold, W.S. The effects of prey size, predator size, and sediment composition on the rate of predation of the blue crab, Callinectes Sapidus Rathbun, on the hard clam, Mercenaria mercenaria (Linné). J. Exp. Mar. Biol. Ecol. 1984, 80, 207–219. [Google Scholar] [CrossRef]
- Eggleston, D.B. Foraging behavior of the blue crab, Callinectes sapidus, on juvenile oysters, Crassostrea virginica: effects of prey density and size. Bull. Mar. Sci. 1990, 46, 62–82. [Google Scholar]
- Lin, J. Predator-prey interactions between blue crabs and ribbed mussels living in clumps. Estuar. Coast Shelf Sci. 1991, 2, 61–69. [Google Scholar] [CrossRef]
- Prado, P.; Peñas, A.; Ibáñez, C.; Cabanes, P.; Jornet, L.; Álvarez, N.; Caiola, N. Prey size and species preferences in the invasive blue crab, Callinectes sapidus: Potential effects in marine and freshwater ecosystems. Estuar. Coast. Shelf Sci. 2020, 245, 106997. [Google Scholar] [CrossRef]
- Haddon, M.; Wear, R.G.; Packer, H.A. Depth and density of burial by the bivalve Paphies ventricosa as refuges from predation by the crab Ovalipes catharus. Mar. Biol 1987, 94, 25–30. [Google Scholar] [CrossRef]
- Sponaugle, S.; Lawton, P. Portunid crab predation on juvenile hard clams: effects of substrate type and prey density. Mar. Ecol. Progr. Ser. Oldendorf 1990, 67, 43–53. [Google Scholar]
- Seitz, R.D.; Lipcius, R.N.; Seebo, M.S. Food availability and growth of the blue crab in seagrass and unvegetated nurseries of Chesapeake Bay. J. Exp. Mar. Biol. Ecol 2005, 319, 57–68. [Google Scholar] [CrossRef]
- Gosling, E. Marine Bivalve Molluscs, 2nd edn. West Sussex UK Wiley Blackwell. 2015, West Sussex, UK. [Google Scholar]
- Fraser, S.; Shelmerdine, R.L.; Mouat, B. (2018). Razor clam biology, ecology, stock assessment, and exploitation: a review of Ensis spp. in Wales. UHI Research Database pdf download summary, 2018; Welsh Government, 2018; pp. 62. [Google Scholar]
- Castejón, D.; Guerao, G. (2013). A new record of the American blue crab, Callinectes sapidus Rathbun, 1896 (Decapoda: Brachyura: Portunidae), from the Mediterranean coast of the Iberian Peninsula. BioInvasions Rec. 2013, 2, 141–143. [Google Scholar] [CrossRef]
- López, V.; Rodon, J. , Diagnosi i situació actual del cranc blau (Callinectes sapidus) al Delta de l’Ebre. Informe Tècnic-Servei de Recursos Marins, Direcció General de Pesca i Afers Marítims, Generalitat de Catalunya, 2018; 86 pp.
- López, V. Seguiment del cranc blau (Callinectes sapidus) al Delta de l’Ebre. Informe Tècnic-Servei de Recursos Marins, Direcció General de Pesca i Afers Marítims, Monverte Estudis Ambientals, Amposta, 2020; 127 pp.
- Baeta, M.; Solís, M.A.; Frias-Vidal, S.; Claramonte, L.; Ballesteros, M. Management and ecology of the wedge clam (Donax trunculus) in the NW Mediterranean Sea: The case of Ebro Delta (NE Spain). Reg. Stud. Mar. Sci. 2023, 66, 103158. [Google Scholar] [CrossRef]
- Blundon, J.A.; Kennedy, V.S. (1982a). Refuges for infaunal bivalves from blue crab, Callinectes sapidus (Rathbun), predation in Chesapeake Bay. J. Exp. Mar. Biol. Ecol. 1982, 56, 67–81. [Google Scholar] [CrossRef]
- Muir, S.D. The biology of razor clams (Ensis spp.) and their emergent fishery on the West coast of Scotland. PhD thesis, University of London. 2003; pp. 280.
- Blundon, J.A.; Kennedy, V.S. Mechanical and behavioral aspects of blue crab, Callinectes sapidus (Rathbun), predation on Chesapeake Bay bivalves. J. Exp. Mar. Biol. Ecol. 1982, 65, 47–65. [Google Scholar] [CrossRef]
- Ebersole, E.L.; Kennedy, V.S. Prey preferences of blue crabs Callinectes sapidus feeding on three bivalve species. Mar. Ecol. Progr. Ser. 1995, 118, 167–177. [Google Scholar] [CrossRef]
- Kutluyer Kocabaş, F.; Kocabaş, M.; Çanakçi, A.; Karabacak, A.H. Mechanical property and structural-elemental analysis of marine bivalve mollusc shells: Cerastoderma edule, Chamelea gallina, Donax trunculus, Ruditapes decussatus. Internat. Aquat. Res. 2023, 15, 39–50. [Google Scholar]
- Bejaoui, S.; Rabeh, I.; Chetoui, I.; Telahigue, K.; Ghribi, F.; Fouzai, C.; El Cafsi, M. Examination of the nutritional value of four bivalves species from Bizerte lagoon. INSTM Bull. Mar. Freshw. Sci. 2019, 46, 71–79. [Google Scholar]
- Carrasco, N.; Hine, P.M.; Durfort, M.; Andree, K. B.; Malchus, N.; Lacuesta, B.; Gonzalez, M.; Roque, A.; Rodgers, R.; Furones, M.D. Marteilia cochillia sp. nov., a new Marteilia species affecting the edible cockle Cerastoderma edule in European waters. Aquaculture. 2013, 412, 223–230. [Google Scholar] [CrossRef]
- Baeta, M.; Rubio, C.; Breton, F. (2021). Impact of mechanized clam dredging on the discarded megabenthic fauna on the Catalan coast (NW Mediterranean). J. Mar. Biol. Assoc. UK. 2021, 101, 545–553. [Google Scholar] [CrossRef]
- Piersma, T.; Koolhaas, A.; Dekinga, A.; Beukema, J.J.; Dekker, R.; Essink, K. Long-term indirect effects of mechanical cockle-dredging on intertidal bivalve stocks in the Wadden Sea. J. Appl. Ecol. 2001, 38, 976–990. [Google Scholar] [CrossRef]
- Ramón, M.; Cano, J.; Peña, J.B.; Campos, M.J. Current status and perspectives of mollusc (bivalves and gastropods) culture in the Spanish Mediterranean. Bolet. Instit. Esp. Oceanograf 2005, 21, 361–373. [Google Scholar]
- Köck, M.; Farré, M.; Martínez, E.; Gajda-Schrantz, K.; Ginebreda, A.; Navarro, A.; López de Alda, M.; Barceló, D. Integrated ecotoxicological and chemical approach for the assessment of pesticide pollution in the Ebro River delta (Spain). J. Hydrol, 2010; 383, 73–82. [Google Scholar]
- De Juan, S.; Demestre, M.; Sanchez, P. Exploring the degree of trawling disturbance by the analysis of benthic communities ranging from a heavily exploited fishing ground to an undisturbed area in the NW Mediterranean. Sci. Mar. 2011, 75, 507–516. [Google Scholar] [CrossRef]
- Nerot, C.; Lorrain, A.; Grall, J.; Gillikin, D.P.; Munaron, J.M.; Le Bris, H.; Paulet, Y.M. Stable isotope variations in benthic filter feeders across a large depth gradient on the continental shelf. Estuar. Coast. Shelf Sci. 2012, 96, 228–235. [Google Scholar] [CrossRef]
- Prado, P.; Baeta, M.; Mestre, E.; Solis, M.A.; Sanhauja, I.; Gairin, I.; Camps-Castellà, J.; Falco, S.; Ballesteros, M. Trophic role and predatory interactions between the blue crab, Callinectes sapidus, and native species in open waters of the Ebro Delta. Estuar. Coast. Shelf Sci. 2024, 298, 108638. [Google Scholar] [CrossRef]
Figure 1.
Blue crab-predated individuals of each bivalve species. (a) Ruditapes spp., (b) C. glaucum; and (c) E. siliqua.
Figure 1.
Blue crab-predated individuals of each bivalve species. (a) Ruditapes spp., (b) C. glaucum; and (c) E. siliqua.

Figure 2.
Results of blue crab predation experiments with the different species of bivalves. (a) Multiple preference with sand; (b) Multiple preference without sand; (c) No choice with sand; (d) No choice without sand. Errors are SE.
Figure 2.
Results of blue crab predation experiments with the different species of bivalves. (a) Multiple preference with sand; (b) Multiple preference without sand; (c) No choice with sand; (d) No choice without sand. Errors are SE.
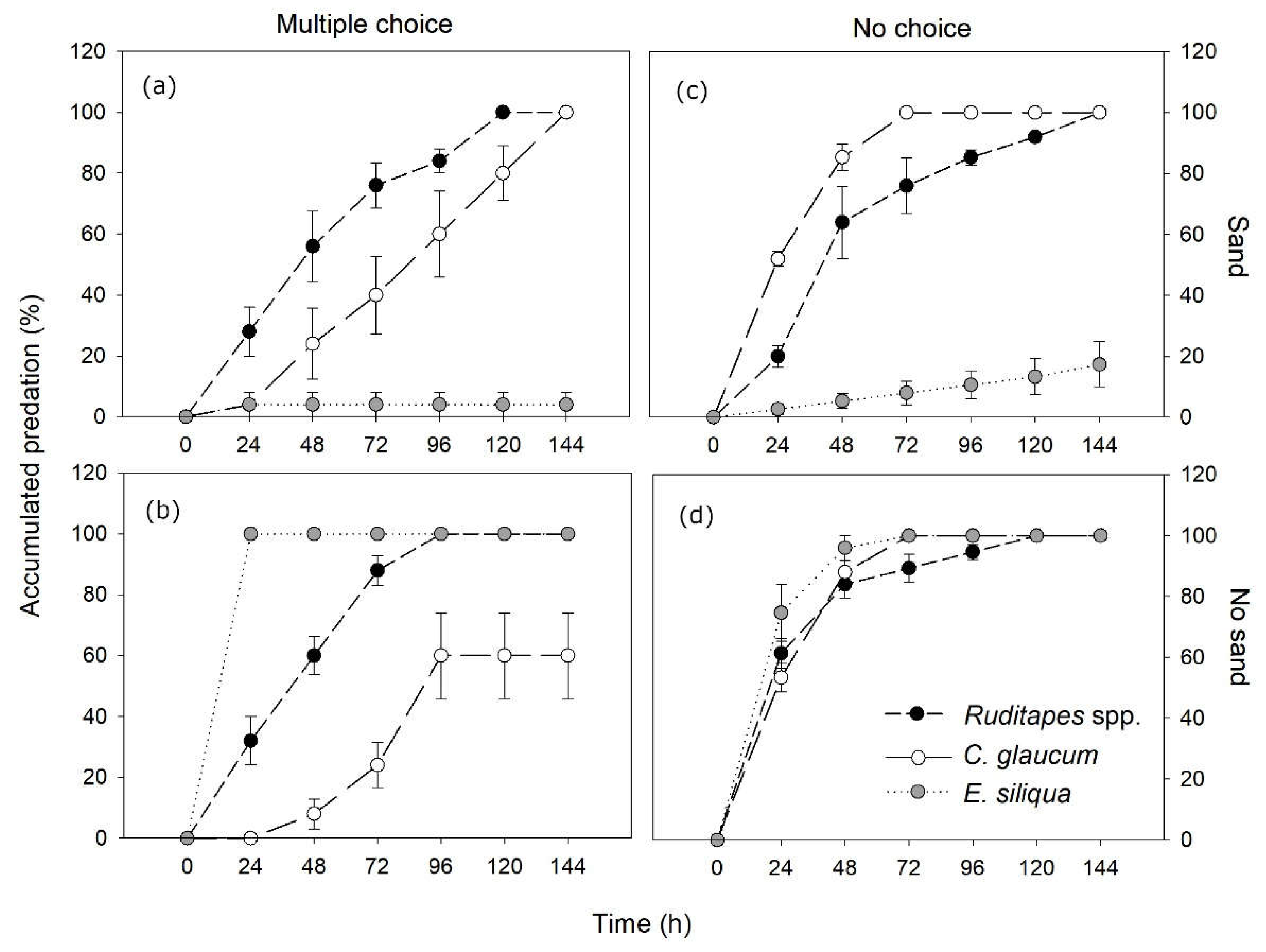
Figure 3.
Fisheries trends of the main bivalve species in the Ebro Delta (cockles, Cerastoderma spp.; the clams, Ruditapes spp.; the razor clams, E. siliqua, and S. marginatus, the wedge clam Donax spp., and several other species of Venus clams) before and after the beginning of blue crab captures in 2016 (dashed line).
Figure 3.
Fisheries trends of the main bivalve species in the Ebro Delta (cockles, Cerastoderma spp.; the clams, Ruditapes spp.; the razor clams, E. siliqua, and S. marginatus, the wedge clam Donax spp., and several other species of Venus clams) before and after the beginning of blue crab captures in 2016 (dashed line).

Table 1.
Results of the 2-way repeated measures ANOVA of cumulative predation rates bivalve species (Ruditapes spp., Cerastoderma glaucum, and Ensis siliqua) using two different substrates (with and without sand). A) Multiple choice results with bivalve species offered simultaneously to each blue crab individual. B) No choice results for each bivalve species. Statistically significant results are indicated in bold.
Table 1.
Results of the 2-way repeated measures ANOVA of cumulative predation rates bivalve species (Ruditapes spp., Cerastoderma glaucum, and Ensis siliqua) using two different substrates (with and without sand). A) Multiple choice results with bivalve species offered simultaneously to each blue crab individual. B) No choice results for each bivalve species. Statistically significant results are indicated in bold.
| A) Multiple choice experiments | df | MS | F | P | Eta square |
|---|---|---|---|---|---|
| Between subjects | |||||
| Time (Ti) | 4.33 | 32677.58 | 196.38 | 0.000 | 0.891 |
| Ti x S | 4.33 | 1568.55 | 9.42 | 0.000 | 0.282 |
| Ti x Sp | 8.66 | 4207.57 | 25.28 | 0.000 | 0.678 |
| Ti x S x Sp | 8.66 | 1915.41 | 11.51 | 0.000 | 0.490 |
| Error | 103.98 | 166.39 | |||
| Within subjects | |||||
| Substrate (S) | 1 | 31697.14 | 42.12 | 0.000 | 0.637 |
| Species (Sp) | 2 | 15716.19 | 20.88 | 0.000 | 0.635 |
| S x Sp | 2 | 45274.28 | 60.17 | 0.000 | 0.834 |
| Error | 24 | 752.38 | |||
| B) No choice experiments | df | MS | F | P | Eta square |
| Between subjects | |||||
| Time (Ti) | 3.57 | 50233.04 | 577.15 | 0.000 | 0.960 |
| Ti x S | 3.57 | 2235.88 | 25.68 | 0.000 | 0.517 |
| Ti x Sp | 7.13 | 1839.54 | 21.13 | 0.000 | 0.638 |
| Ti x S x Sp | 7.13 | 1607.84 | 18.47 | 0.000 | 0.606 |
| Error | 85.67 | 87.03 | |||
| Within subjects | |||||
| Substrate (S) | 1 | 44200.84 | 217.32 | 0.000 | 0.901 |
| Species (Sp) | 2 | 19663.06 | 96.67 | 0.000 | 0.890 |
| S x Sp | 2 | 26469.41 | 130.14 | 0.000 | 0.916 |
| Error | 24 | 203.38 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Copyright: This open access article is published under a Creative Commons CC BY 4.0 license, which permit the free download, distribution, and reuse, provided that the author and preprint are cited in any reuse.
Effects of Sand Burial Depth on Bivalve Species Predation by the Invasive Blue Crab, Callinectes sapidus
Patricia Prado
et al.
,
2024
Decapods Associated with the Southern King Crab (Lithodes santolla) Fishery in Central Patagonia (44º S, Chile).
Guillermo Figueroa-Muñoz
et al.
,
2021
MDPI Initiatives
Important Links
© 2024 MDPI (Basel, Switzerland) unless otherwise stated









